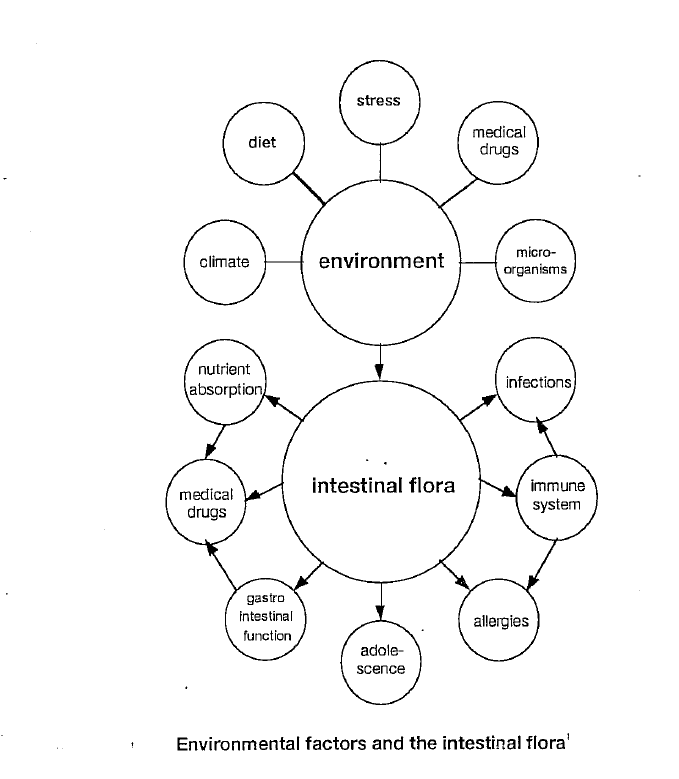
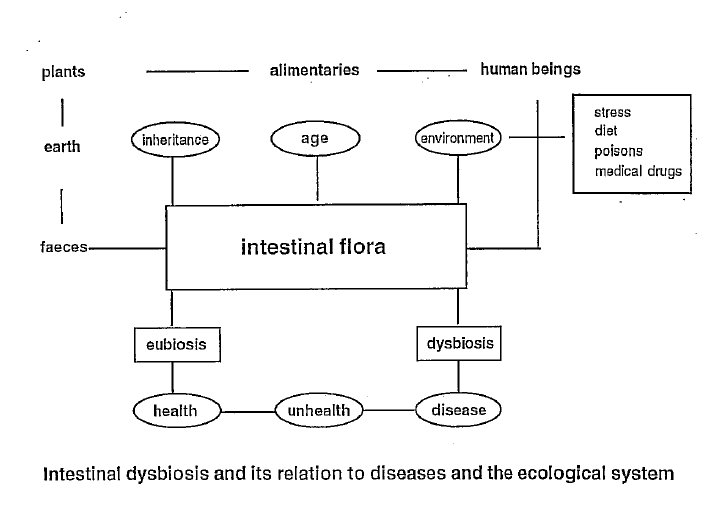
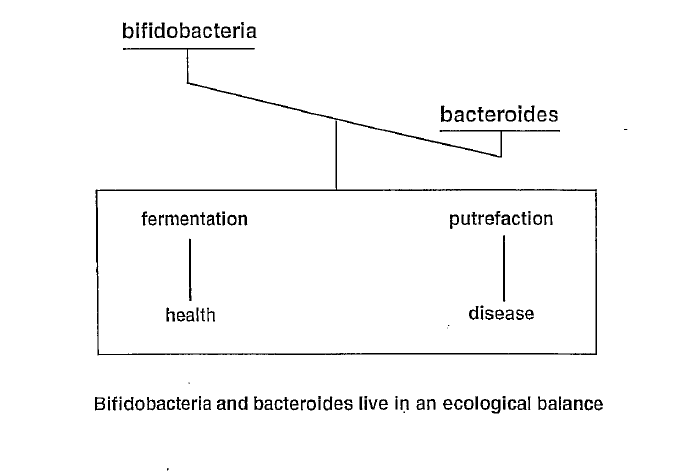
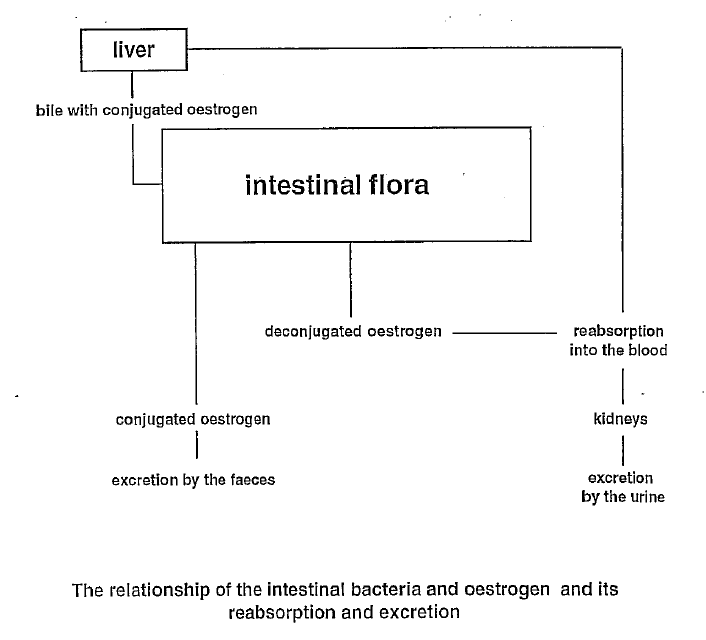
The lymphatic system consists of two parts:
- The lymph ducts
- The lymph nodes
The lymphatic system is a constituent part of the vascular system of the entire body. The lymph vessels begin as “blind channels”: that is, with closed ends in the organs and tissues. Lymph vessels have endothelial openings through which tissue fluid and small particles can flow. As they pass through the body, these microscopically small lymph capillaries become larger vessels, which are eventually provided with valves, similar to the veins. A distinction can be made here between the superficial lymph ducts — which can be recognized as red lines, as during an inflammation — and the deeper lymph channels.
Many of the deeper lymph vessels merge into the superficial ducts in the course of their passage.
The lymph nodes appear at long and short intervals in the lymph ducts. These nodes function as filters. As a result, the lymph vessels and the passage of lymph are interrupted once or several times in their progress toward the heart. At some places, there is a long interval between the individual lymph-node stations. At others, this interval is quite short. In some areas, the lymph nodes are thinly distributed; at others, there is a considerable density of them. From the anatomical standpoint, distinction can be made among the four main areas of the body in which the lymph nodes are located with considerable concentration:
- The region of the head
- The region of the throat and neck
- The upper half of the trunk and the upper extremities
- The lower half of the trunk and the lower extremities.
The lymph vessels function in a manner similar to that of the veins. Contractions of the body’s muscles and the functioning of the valves in the lymph ducts move the lymph onward until it flows into the venous system, in the region of the angulus venosus.
B. Physiology and pathophysiology
The exchange of substances between blood and tissue takes place in the capillary area. This exchange depends on several factors — among them, the difference in concentration between intra- and extravascular substances. Expressed simply, the capillary walls represent a type of ultra-low-penetration filter, which is easily permeated by water and crystalline dissolved substances. For colloids, however, these walls are practically impermeable.
A constant process of filtration and reabsorption takes place in such vessels. This dynamic system is characterized most significantly by the oncotic pressure of the liquids involved, and by the transmural pressure of the capillary membranes.
In general, and under normal circumstances, the filtration and the reabsorption process do not take place at the same rates, and the filtration process will predominate. In addition, the proteins which pass into the interstitial liquid cannot return to the capillaries from which they came. Water and protein would therefore increasingly accumulate in the interstitium if they were not returned to the blood vessel system by the lymphatic system.
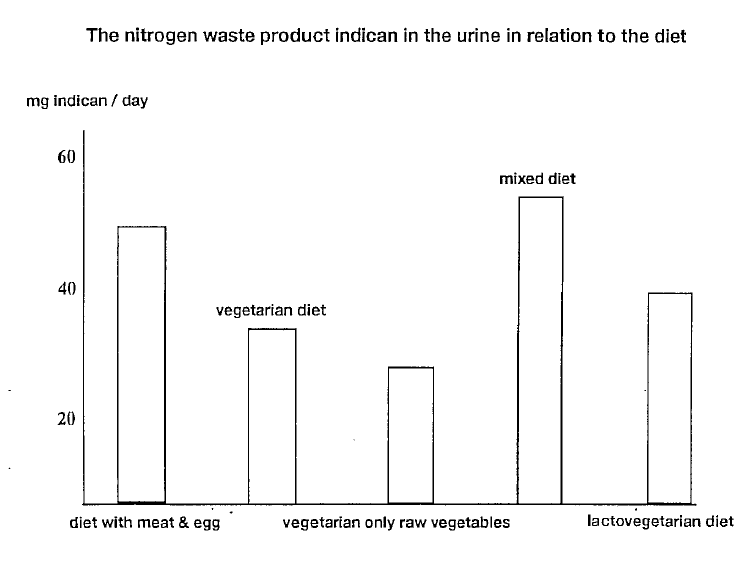
The composition of lymph is very similar to that of blood plasma, with the exception of the lower content of protein in lymph. The protein content of lymph varies over the different regions of the body. Approximately two to three litres of lymph are produced in the body every day.
Fluid will accumulate in the form of edema if there is a disturbance in the relationship between filtration in capillary area, on the one hand, and reabsorption, on the other. This takes place primarily as a concequence of the following:
Formation of edema:
- Obstruction in the lymph ducts
- Hypoalbuminemia
- Increased capillary permeability
- Restriction in drainage in the lymph ducts in cases of increased venous pressure
The most frequent causes of these phenomena can be found in the following illnesses:
The most frequent causes of edema development:
- Cardiac insufficiency with congestion of venous blood
- Kidney diseases with protein loss
- Hypertension (increased pressure; rise in capillary blood pressure)
- Inflammatory disorders (increased capillary permeability)
- Obstruction of lymph passage by congested lymph nodes (inflammation, tumor debris, surgery, etc.)
Edema first becomes visible when the extravascular liquid volume has increased by approximately thirty percent. Circadian fluctuations can occur, among the healthy as well, and they should be considered as physiological in nature.
C. Therapy
Classification of lymphedema
Stage 1: The latency and interval stage
Stage 2: Reversible lymphedema
Stage 3: Irreversible lymphedema
Stage 4: Lymphostatic elephantiasis
If lymphedema is not promptly treated, it worsens in the sense of a vicious circle. Owing to the high content of protein in the extravascular area, the entire afflicted region is also very susceptible to infection. Extremely minor injuries can lead to serious complications. Physicians have also published accounts that cirrhosis can develop as a result of such circumstances in connecting tissues. Functional restriction can therefore result, and motor and sensory paralysis can ensue as a consequence of the enclosure of nerves.
The therapy of lymphadema should primarily be directed toward the primary disease. Supplementary therapy should, however, include manual lymph drainage and the administration of Lymphomyosot®
Manual lymph drainage should be performed only by specially trained therapists, with care taken that the edematous tissue not be damaged. The pressure applied in manual lymph drainage should therefore never exceed the empirically obtained value of thirty-seven …….
Since relief of obstructions by manual methods necessitates therapy over extremely long periods of time, inquiries for an alternative in the form of medication have naturally arisen. Lymphomyosot® can be recommended as an effective basic therapy for such disorders.
Lymphomyosot® has a channelling, draining effect and promotes mesenchymal detoxification. Throughout therapy directed toward the lymph system, it is very important that lymph drainage never be completely interrupted — a development which would lead to worsening of the lymphedema. This requirement, however, indirectly confirms the effectiveness of Lymphomyosot® in its mesenchymal, detoxifying, and draining effects.
To assess the effects of Lymphomyosot®, I would like to present several findings in very brief form.
- Zimmermann investigated the effects of Lymphomyosot® on patients with venous leg edema. Freedom from symptoms was achieved over a period of one to five weeks by administration of one ampule of Lymphomyosot® twice a week. The patients tolerated the medication outstandingly well.
- Hummel recommends a dosage of thirty drops three times a day over a period of several months, for edematous congestive eczema of the legs.
- Kirchhoff compared the effects of administering Lymphomyosot® ampules with the results of manual lymph drainage, over a period of five weeks, for patients with arm lymphedema following breast amputation. His conclusions were that Lymphomyosot® and manual lymph drainage were equally effective. His results also showed that a combination of both therapeutic measures produced greater effectiveness than either alone.
An additional major area of application for Lymphomyosot® is the precancerous state. This condition is not entirely restricted to actual precancerous disorders alone: it also includes a complec of symptoms which in recent times has caused a great deal of apprehension among practicing pysicians.
The precancerous state
- Excessive fatigue
- Insomnia
- Incipient loss of weight
- Nervousness and depression
- Constipation
- Impaired peripheral circulation of blood
- Multiple nevi
- Moderate elevation of the erythrocyte sedimentation rate (ESR)
These symptoms can lead — in the sense of Hans Heinrich Reckeweg’s homotoxicology — to the assumption of a retoxic phenomenon under the influence of an impregnation phase. These symptoms can be effectively treated by basic therapy as developed by Leimbach.
Leimbach’s program of therapy for the precancerous state
AP.
- Lymphomyosot®
- Psorinoheel®
- Galium-Heel®
- Phosphor-Homaccord® aa. ad 120.0
- MDS, 3 times daily, 40 drops a.c.
Also very effective here is initial administration by injection of Ubichinon Compositum and Coenzyme Compositum, for catalytic stimulation of organelle functions. It is also equally important to assure complete case-history recording of the preliminary phases of lymphedema, and to administer the corresponding nosode preparations.
Pure homeopathic therapy will not of course alone cure a malignant disease. Primary treatment must consist of classical cancer therapy as developed by modern scientific medicine.
Nevertheless, the helping hand of homeopathy can indeed provide effective supplementary support as the fourth column of cancer treatment — in addition to surgery, radiology, and chemotherapy.
Owing to its reliable draining and channeling effects, Lymphomyosot® should be administered in the pre- and post-operative phases of cancer therapy.
In such pre- and post-surgical stages, Mayer-Langsdorff has been successful in combination administration of the following HEEL preparations, in addition to Lymphomyosot®: Psorinoheel®, Galium-Heel®, and Phosphor-Homaccord®.
In the Heidelberg Special Clinic for Thorax Surgery, Winterberg has also effectively treated bronchial cancer patients for years with long-term administration of Lymphomyosot®, Galium-Heel®, Traumeel®, and Bronchalis-Heel®.
As the above-stated publications have attested, the physician can therefore rely on the fact that Lymphomyosot® will effectively promote lymph drainage.
The techniques of lymph drainage, however, have remained a neglected area of medical therapy, at least among the modern physicians of West Germany. The same neglect has also been suffered by venous therapy — a field of treatment which can also be effectively supported by administration of Lymphomyosot®.
Professor Dr. Volker Wienert, of the University of Aachen, who holds the only West German professorship for dermatological phlebology, once made the following statement on the significance of venous therapy among modern physicians:
“Veins have a poor image among modern scientists since, after all, they must be included among the bodily organs of disposal. And it is well known that the functions of disposal systems have been generally neglected.”
The lymphatic system of course plays a primary role in the body’s disposal system. In the hopes that you will accord the body’s disposal system the significance which it critically deserves, I thank you for your attention to my remarks.